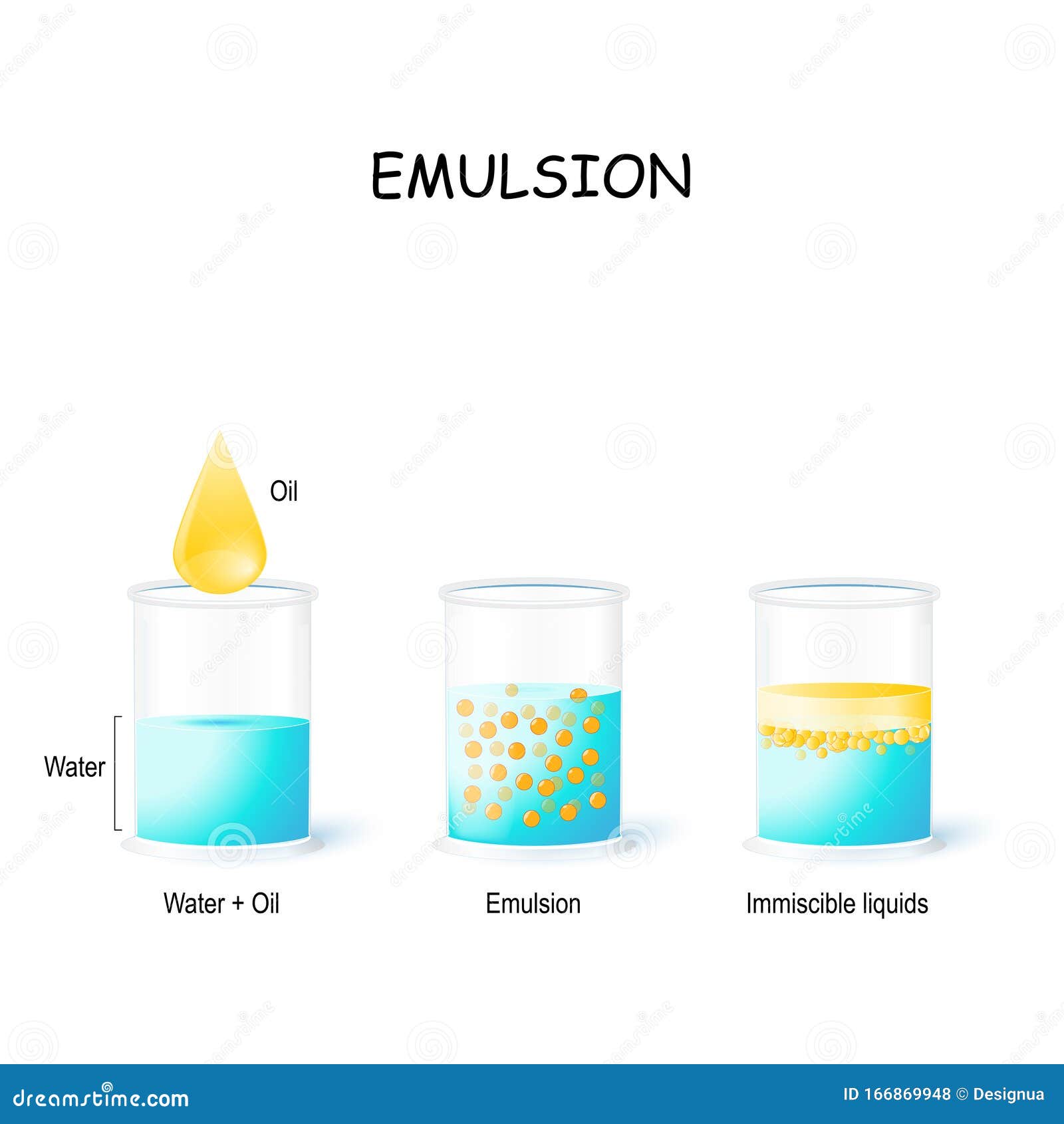
There are three common types of instabilities that occur in emulsions. Most emulsions aren’t stable unless an emulsifier is added to the mixture. The ability of an emulsion to retain its form is called its stability. To stop the emulsion from breaking apart, emulsifiers or emulsifying agents are used. This is called ‘cracking of emulsion’ or ‘phase inversion’. If an emulsion is left to rest, it will start to disintegrate. The two liquid phases do disperse in each other when shaken, but most emulsions won’t stay that way for long. The kind of emulsion that results due to mixing of two liquids is largely dependent on the type of emulsifier used and the volume of the two liquids.Įmulsions are unstable. Water-in-oil: If the aqueous phase is immersed in the continuous oil phase, then it is a water-in-oil emulsion.Oil-in-water: If the aqueous phase is the continuous phase and the oil phase is dispersed, then it’s an oil-in-water emulsion.The negative reel of photographic film is layered with an emulsion of silver halide in gelatin.Įmulsions can be classified into two types.Mayonnaise is an oil-in-water emulsion, with lecithin in the egg yolk stabilizing the emulsion.Butter is an emulsion of water and fat.Crema put on espressos and coffees is an emulsion of water and coffee oil.Egg yolk is an emulsion containing the emulsifying agent lecithin. 
Oil and water mixtures, one of the most common examples, are emulsions when shaken together.Other natural emulsions are egg yolk, butter and mayonnaise.Put simply, an emulsion is basically a mixture of liquids that doesn’t entirely ‘mix’ completely. Milk itself is a naturally occurring emulsion a dispersion of fats and water. The process of making an emulsion is known as emulsification.Emulsion is derived from the Latin word ‘mulego’, which means ‘to milk’. An emulsion is formed when two immiscible liquids are mixed together through vigorous shaking. A subset of the larger group of two-phased systems, emulsions is the term usually given to a liquid system of two immiscible liquid (liquids that don’t mix together) phases, in which one phase is dispersed in the other.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
